Header Skipped.
Defining recovery neurobiology of injured spinal cord by synthetic matrix-assisted hMSC implantation
Implantation of adult human bone marrow-derived stromal stem cells in synthetic scaffold restored walking in rats with acute spinal cord injury, improved sensory function, and reduced major complications of bladder dysfunction and pain.
Highlights:
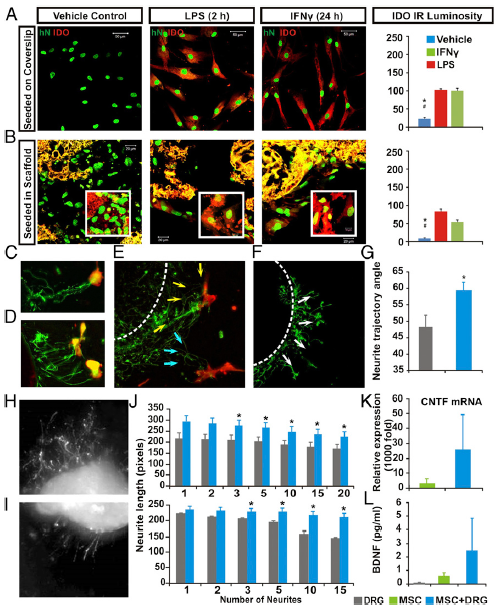 Legend: Multimodal effects of hMSCs in vitro. Compared with saline control, LPS or IFN exposure significantly augmented IDO expression (red) in both (A) non-scaffolded hMSCs and (B) PLGA-scaffolded hMSCs, as shown by human nuclei (hN) immunostaining (green; P < 0.05: *, control vs. LPS; #, control vs. IFN-γ; n = 5; one-way ANOVA with Tukey’s post hoc test). PLGA scaffolds showed yellow autofluorescence under dual channels. (C–E) DRG in organotypic coculture grew neurites (GAP43+, green) that track toward nearby hMSCs (CD90+, red; arrows), compared with (F) a more radial neurite pattern in DRG cultured with scaffold only (G). There were significantly more angular neurite paths in DRG and scaffolded hMSC cocultures (59.25 ± 2.9°) relative to the DRG and scaffold-alone group (48.15 ± 4.1°; P = 0.026, Student’s t test). Images showed different lengths of neurite outgrowth at the (H) distal and (I) proximal sites of axotomized DRG cocultured with PLGA-scaffolded hMSCs or scaffold alone, respectively. (J, Upper) The scaffolded hMSC and DRG cocultures had significant increases in mean total lengths when 3–20 neurites per DRG were averaged (*P = 0.032, n = 6, Mann–Whitney) and also (J, Lower) significantly increased the maximum absolute length of DRG neurite outgrow when 3–15 neurites per DRG were assessed (*P = 0.026, n = 6, Mann–Whitney). (K) Relative CNTF mRNA expression in scaffolded hMSCs was significantly elevated, as was (L) secretion of human BDNF (P < 0.05, one-way ANOVA) in the scaffolded hMSC + DRG coculture system.
Legend: Multimodal effects of hMSCs in vitro. Compared with saline control, LPS or IFN exposure significantly augmented IDO expression (red) in both (A) non-scaffolded hMSCs and (B) PLGA-scaffolded hMSCs, as shown by human nuclei (hN) immunostaining (green; P < 0.05: *, control vs. LPS; #, control vs. IFN-γ; n = 5; one-way ANOVA with Tukey’s post hoc test). PLGA scaffolds showed yellow autofluorescence under dual channels. (C–E) DRG in organotypic coculture grew neurites (GAP43+, green) that track toward nearby hMSCs (CD90+, red; arrows), compared with (F) a more radial neurite pattern in DRG cultured with scaffold only (G). There were significantly more angular neurite paths in DRG and scaffolded hMSC cocultures (59.25 ± 2.9°) relative to the DRG and scaffold-alone group (48.15 ± 4.1°; P = 0.026, Student’s t test). Images showed different lengths of neurite outgrowth at the (H) distal and (I) proximal sites of axotomized DRG cocultured with PLGA-scaffolded hMSCs or scaffold alone, respectively. (J, Upper) The scaffolded hMSC and DRG cocultures had significant increases in mean total lengths when 3–20 neurites per DRG were averaged (*P = 0.032, n = 6, Mann–Whitney) and also (J, Lower) significantly increased the maximum absolute length of DRG neurite outgrow when 3–15 neurites per DRG were assessed (*P = 0.026, n = 6, Mann–Whitney). (K) Relative CNTF mRNA expression in scaffolded hMSCs was significantly elevated, as was (L) secretion of human BDNF (P < 0.05, one-way ANOVA) in the scaffolded hMSC + DRG coculture system.
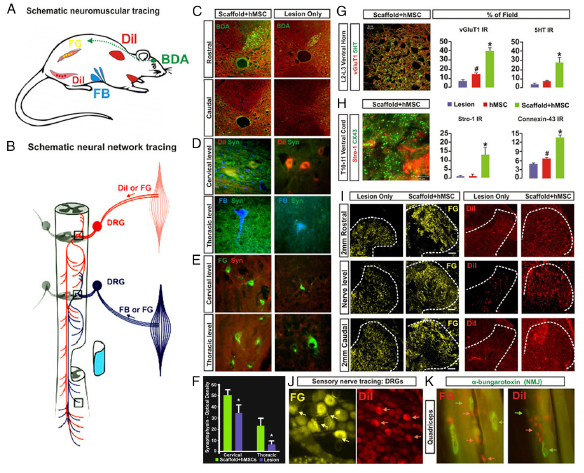 Legend: Neurobiological benefits resulting from the scaffolded hMSC treatment. (A) Tracing regimens: BDA (green; injected into motosensory cortex contralateral to hemisection) for tracing the CST on the lesioned side, whereas DiI, Fast Blue (FB), and Fluoro-Gold (FG) were injected ipsilaterally into cervical region muscles and intercostal and abdominal muscles (Materials and Methods) to trace both primary proprioceptive axons that synapse with the PSN interneurons and with ventral horn motor neurons. DiI and FG were also injected into hindlimb muscles for retrograde tracing of lumbar motor neurons and primary afferent fibers. (B) Schematic of a simplified PSN network consisting of cross-segmental 1° and 2° neurites that project bilaterally and bidirectionally. (C) BDA IR was detected in the ipsilateral CST rostral to the injury epicenter, but not at any levels caudal to the lesion site in both scaffolded hMSC-treated and lesion-only spinal cords, suggesting no CST regeneration. (D and E) Images of DiI-, FB-, or FG-traced cervical and thoracic ventral horn motor neurons, respectively, and the synapses in the surrounding regions that were costained by antibody against synaptophysin (Syn, green), a synaptic vesicle marker. Scaffolded hMSCs-treated spinal cords showed much higher Syn IR density in both cervical and thoracic cord regions, compared with lesion-only (or hMSC sonly) controls (F; P < 0.05, paired Student’s t test, n = 5 per group). (G, Left) Tissue slices selected from upper lumbar segment 5 mm caudal to the epicenter were immunostained for 5-HT (red) and vGluT1 (green). A representative z-stack confocal plane (20 × 0.1 μm optical sections) of the Rexed Lamina VIII area is shown (Inset) at 600× magnification, and presents typical details of the pericellular distribution of axon terminal vGluT1 and beaded varicosities of 5HT. (Scale bar: 5 μm.) (G, Right) Group average IR of vGluT1 (red) that marks primary proprioceptive terminals and 5HT (green) in lumbar Rexed Lamina VIII were significantly stronger in scaffolded hMSC-treated spinal cords compared with controls. (H) Coimmunostaining of STRO-1 (red), an hMSC marker and Cx43 (green), a gap junction protein, revealed significantly higher signal density of Cx43 in the host ventral cord region of sections immediately caudal to the scaffolded hMSC implant site, compared with the control groups (*, scaffolded hMSC vs. hMSC only and lesion only; #, hMSC only vs. lesion only; P < 0.05, n = 5 per group; one-way ANOVA with Tukey’s post hoc test). Cx43 was expressed mainly by host cells (i.e., STRO-1–negative cells). (I) Retrograde tracing with FG or DiI via T6–T8 intercostal nerve showed FG+ or DiI+ primary afferent fibers and (J) DRG neurons. Scaffolded hMSC treatment drastically increased densities and area of both tracers, relative to the lesion controls, and DRG neuron integrity (J). (K) Motor nerve endings are in close contact with α-bungarotoxin+ nicotinic receptors in the muscles, showing near normal morphology of NMJs in the quadriceps of scaffolded hMSC-treated rats.
Legend: Neurobiological benefits resulting from the scaffolded hMSC treatment. (A) Tracing regimens: BDA (green; injected into motosensory cortex contralateral to hemisection) for tracing the CST on the lesioned side, whereas DiI, Fast Blue (FB), and Fluoro-Gold (FG) were injected ipsilaterally into cervical region muscles and intercostal and abdominal muscles (Materials and Methods) to trace both primary proprioceptive axons that synapse with the PSN interneurons and with ventral horn motor neurons. DiI and FG were also injected into hindlimb muscles for retrograde tracing of lumbar motor neurons and primary afferent fibers. (B) Schematic of a simplified PSN network consisting of cross-segmental 1° and 2° neurites that project bilaterally and bidirectionally. (C) BDA IR was detected in the ipsilateral CST rostral to the injury epicenter, but not at any levels caudal to the lesion site in both scaffolded hMSC-treated and lesion-only spinal cords, suggesting no CST regeneration. (D and E) Images of DiI-, FB-, or FG-traced cervical and thoracic ventral horn motor neurons, respectively, and the synapses in the surrounding regions that were costained by antibody against synaptophysin (Syn, green), a synaptic vesicle marker. Scaffolded hMSCs-treated spinal cords showed much higher Syn IR density in both cervical and thoracic cord regions, compared with lesion-only (or hMSC sonly) controls (F; P < 0.05, paired Student’s t test, n = 5 per group). (G, Left) Tissue slices selected from upper lumbar segment 5 mm caudal to the epicenter were immunostained for 5-HT (red) and vGluT1 (green). A representative z-stack confocal plane (20 × 0.1 μm optical sections) of the Rexed Lamina VIII area is shown (Inset) at 600× magnification, and presents typical details of the pericellular distribution of axon terminal vGluT1 and beaded varicosities of 5HT. (Scale bar: 5 μm.) (G, Right) Group average IR of vGluT1 (red) that marks primary proprioceptive terminals and 5HT (green) in lumbar Rexed Lamina VIII were significantly stronger in scaffolded hMSC-treated spinal cords compared with controls. (H) Coimmunostaining of STRO-1 (red), an hMSC marker and Cx43 (green), a gap junction protein, revealed significantly higher signal density of Cx43 in the host ventral cord region of sections immediately caudal to the scaffolded hMSC implant site, compared with the control groups (*, scaffolded hMSC vs. hMSC only and lesion only; #, hMSC only vs. lesion only; P < 0.05, n = 5 per group; one-way ANOVA with Tukey’s post hoc test). Cx43 was expressed mainly by host cells (i.e., STRO-1–negative cells). (I) Retrograde tracing with FG or DiI via T6–T8 intercostal nerve showed FG+ or DiI+ primary afferent fibers and (J) DRG neurons. Scaffolded hMSC treatment drastically increased densities and area of both tracers, relative to the lesion controls, and DRG neuron integrity (J). (K) Motor nerve endings are in close contact with α-bungarotoxin+ nicotinic receptors in the muscles, showing near normal morphology of NMJs in the quadriceps of scaffolded hMSC-treated rats.